Study objectives
This study describes a cost effective limb preservation protocol for diabetic foot patients indicated for limb amputation in a low income developing country. The protocol uses Multidex® maltodextrin/ascorbic acid dressing (DeRoyal®, Tennessee, USA) as a primary dressing and gauze as a secondary dressing following transmetatarsal amputation.
Diabetic foot challenges in panama
• Diabetes affects 194 million individuals worldwide
• Prevalence of diabetes in Panama is similar to world prevalence (9%)
• Most Panamanian diabetics have limited resources and education and must utilize Panama’s socialized health care, and most require use of feet for employment.
• Panama has a warm humid climate year-round creating difficult conditions to heal diabetic foot wounds.
• A protocol is needed to reduce treatment time and burden on healthcare systems, specially in developing countries, such as Panama.
Introduction: limb preservation following diabetic foot ulceration
• Major limb amputation is required for 7 to 20% of diabetic foot ulcer cases1. Within 3 to 5 years, 58% of these patients require amputation of contralateral limb. Three-year mortality rate is between 20 and 50%.
• Major amputations are estimated to place a US$67.47 million burden on the Panamanian healthcare system annually. Estimation assumes only 7% of diabetic Panamanians require an amputation and does not account for lost wages due to inability to work and reduced quality of life.
• These observations indicate the importance of cost-effective limb preservation to maximize quality of life and life expectancy.
• These motivating factors led to the development of a cost-effective protocol that utilizes sharp debridement, a maltodextrin/ascorbic acid* primary dressing, gauze, and limb offloading.


Methods
• Thirteen patients were included in the study following a transmetatarsal amputation to preserve the limb.
• Wounds dressed daily with a maltodextrin/ascorbic acid based primary dressing and a cotton gauze secondary dressing.
• Appropriate offloading prescribed to each patient.
• Patients followed up once a week for wound assessment and sharp debridement as needed.
• Treatment continued until the wound resolved or the clinician resolved the wound with grafting.
• Wounds were assessed by digital planimetry(PictZar®) to quantify wound healing through wound size reduction and granulation tissue formation. Wound metrics were analyzed by K-M survival curves.

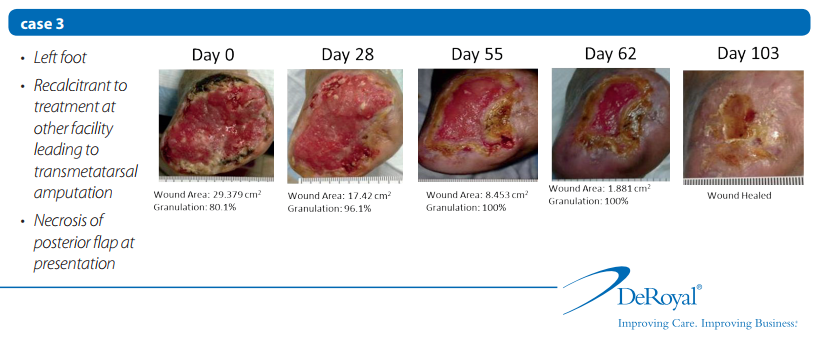
Case 1
Discussion
Twelve of 13 wounds achieved complete healing during the study without skin grafting; the remaining wound achieved 70% healing before skin grafting.
• All wounds achieved complete granulation of wound bed.
85% Median: 27 days; 100% Median: 57 days (range 19 –193).
Allows for grafting procedures if required or preferred.
• Formation of granulation tissue indicates that wound healing is progressing in response to treatment protocol.
• Time to healing has linear relationship with initial wound size. As expected, large wounds required more time to heal.
Study showed a 6.14% healing rate in first 15 weeks.
Survival analysis indicated the median heal time was 103.5 days.
• Time to complete healing of wound is similar to other advance wound care methods, however, far less expensive versus other advanced treatments such as NPWT (Negative Pressure Wound Therapy).
Conclusions
• Effective wound healing demonstrated in this case series of 13 patients utilizing the escribed limb preservation protocol. All limbs were salvaged in the study population.
• Multidex maltodextrin/ascorbic acid* dressing is the key component. It established an ideal moist environment that promotes the body’s natural wound healing process. It also aided in autolytic debridement of dry wounds through use of the gel.
• The components of the limb preservation protocol are cost effective in reducing the burden on the Panamanian healthcare system. Potential cost savings estimated at US$20 million annually if implemented throughout Panama.
US$1200 – $1500 total cost for patients treated with study protocol
Estimated minimal cost with successful NPWT in Panama is US$4800
• The cost savings could be reinvested in diabetes clinical research and in patient education to prevent diabetes and associated complications.
References
1 – Frykberget al. J Foot and Ankle. 2006
2 – World Bank 2012 http://databank.worldbank.org/data/home.aspx
3 – Armstrong et al. The Lancet 366.9498 (2005): 1704 –1710